|
 |
 |
 |
 |
|
|
|






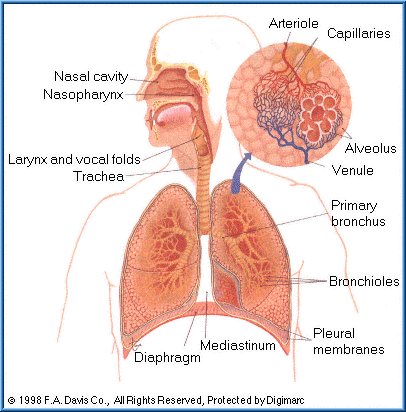
Respiratory Tract

Many environmental and occupational agents as well as some pharmaceuticals are inhaled and enter the respiratory tract. Absorption can occur at any place within the upper respiratory tract. However, the amount of a particular xenobiotic that can be absorbed at a specific location is highly dependent upon its physical form and solubility.
There are three basic regions to the respiratory tract:
 |
 |
 |
|  | nasopharyngeal region
|
 |
|  | tracheobronchial region
|
 |
|  |
pulmonary region.
|
 |
 |
 |
 |
 |
| |

V. C. Scanlon and T. Sanders, Essentials of
Anatomy and Physiology, 2nd edition. F. A. Davis, 1995. |
|
By far the most important site for absorption is the
pulmonary region consisting of the very small airways (bronchioles) and the alveolar sacs of the lung.
The alveolar region has a very large surface area, about 50 times that of the skin. In addition, the alveoli consist of only a single layer of cells with very thin membranes that separate the inhaled air from the blood stream. Oxygen, carbon dioxide and other gases readily pass through this membrane. In contrast to absorption via the gastrointestinal tract or through the skin, gases and particles, which are water-soluble (and thus blood soluble), will be absorbed more efficiently from the lung alveoli. Water-soluble gases and liquid aerosols can pass through the alveolar cell membrane by simple passive diffusion.
In addition to solubility, the ability to be absorbed is highly dependent on the physical form of the agent (that is, whether the agent is a gas/vapor or a particle). The physical form determines penetration into the deep lung.
A gas or vapor can be inhaled deep into the lung and if it has high solubility in the blood, it is almost completely absorbed in one respiration. Absorption through the alveolar membrane is by passive diffusion, following concentration gradient. As the agent dissolves in the circulating blood, it is taken away so that the amount that is absorbed and enters the body may be quite large.
The only way to increase the amount absorbed is to increase the rate and depth of breathing. This is known as ventilation-limitation. For blood-soluble gases, equilibrium between the concentration of the agent in the inhaled air and that in the blood is difficult to achieve.
Inhaled gases or vapors, which have poor solubility in the blood, have quite limited capacity for absorption. The reason for this is that the blood can become quickly saturated. Once saturated, blood will not be able to accept the gas and it will remain in the inhaled air and then exhaled.
The only way to increase absorption would be to increase the rate of blood supply to the lung. This is known as flow-limitation. Equilibrium between blood and the air is reached more quickly for relatively insoluble gases than for soluble gases.
The absorption of airborne particles is usually quite different from that of gases or vapors. The absorption of solid particles, regardless of solubility, is dependent upon particle size.
Large particles (>5 µM) are generally deposited in the nasopharyngeal (head airways region) region with little absorption. Particles 2-5 µM can penetrate into the tracheobronchial region. Very small particles (<1 µM) are able to penetrate deep into the alveolar sacs where they can deposit and be absorbed.
Minimal absorption takes place in the nasopharyngeal region due to the cell thickness of the mucosa and the rapid movement of gases and particles through the region. Within the tracheobronchial region, relatively soluble gases can quickly enter the blood stream. Most deposited particles are moved back up to the mouth where they are swallowed.
Absorption in the alveoli is quite efficient compared to other areas of the respiratory tract. Relatively soluble material (gases or particles) is quickly absorbed into systemic circulation.
Pulmonary macrophages exist on the surface of the alveoli. They are not fixed and not a part of the alveolar wall. They can engulf particles just as they engulf and kill microorganisms. Some non-soluble particles are scavenged by these alveolar macrophages and cleared into the lymphatic system.
Some other particles may remain in the alveoli indefinitely. For example, coal dust and asbestos fibers may lead to black lung or asbestosis, respectively.
The nature of toxicity of inhaled materials depends on whether the material is absorbed or remains within the alveoli and small bronchioles. If the agent is absorbed and is also lipid soluble, it can rapidly distribute throughout the body passing through the cell membranes of various organs or into fat depots. The time to reach equilibrium is even greater for the lipid soluble substances. Chloroform and ether are examples of lipid-soluble substances with high blood solubility.
Non-absorbed foreign material can also cause severe toxic reactions within the respiratory system. This may take the form of chronic bronchitis, alveolar breakdown (emphysema), fibrotic lung disease, and even lung cancer. In some cases, the toxic particles can kill the alveolar macrophages, which results in a lowering of the bodies' respiratory defense mechanism.

  
|
|
|
|